Full Name
ATF2
Long Name Textual
Activating Transcription Factor
Immuno Sequence
GST-ATF2 (19 - 96) [DU 1787]
Mono/Poly clonal
Polyclonal
Gel Image
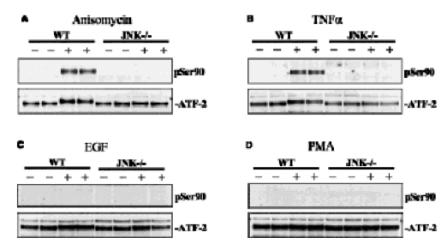
Embryonic fibroblast cells from wildtype (WT) and JNK-/- mice were stimulated for (A) 30 mins with anisomycin (10 μg/ml), (B) for 15 min with TNFalpha (10 ng/ml), (C) for 15 min with EGF (100 ng/ml) and (D) for 30 mins with PMA (100 ng/ml). The cells were lysed and ATF2 was immunoprecipitated from the lysates using 2 μg anti-ATF2 (S512A) per 5 mg of extract. The immunoprecipitate was denatured in SDS, subjected to SDS/PAGE and transferred to nitrocellulose membranes. The membranes were immunoblotted with an antibody that recognises ATF2 phosphorylated at Ser 90 (anti- ATF2 phospho Ser 90 S587A) and with an antibody that recognises the phosphorylated and unphosphorylated forms of ATF2 equally well (anti-ATF2 S512A). Binding of the primary antibody was detected using rabbit peroxidase conjugated anti- sheep IgG antibody (1 in 10, 000 dilution, Pierce) followed by enhanced chemiluminescence (ECL, Amersham).
Sheep No
S512A
Unit Source
Aliquot
0.1mg
Price
£150.00


