Full Name
CRTC3 phospho Ser 370
Long Name Textual
CREB regulated transcription coactivator 3
Synonyms
CRTC3
Immuno Sequence
RLFSLS*NPSLST [residues 365 - 376 of human]
Mono/Poly clonal
Polyclonal
Gel Image
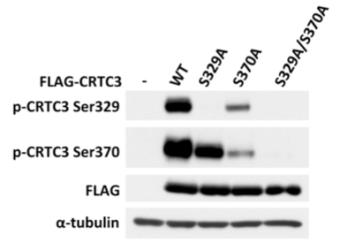
Constructs encoding wild-type Flag-CRTC3 and FLAG-CRTC3 with relevant phosphosites mutated to Ala were expressed in HEK293 cells. Cell extract protein (25 ug) was separared by SDS-Page and immunoblotted with anti-CRTC3 phospho Ser 329 (S256D) or anti-CRTC3 phospho Ser 370 (S253D) at 125 ng/ml in the presence of 1.25 ug/ml of the appropriate non-phospho peptide. Expression of each CRTC3 construct was verified by blotting using anti-FLAG. Antibodies raised against phospho Ser 329 and phospho Ser 370 detect overexpressed wildtype CRTC3 by immunoblot. The antibody against phospho Ser 329 failed to detect CRTC3 with Ser329 mutated to Ala. The antibody against phospho Ser 370 weakly detected CRTC3 when Ser 370 was mutated to Ala but did not recognise CRTC3 with Ser 329 and Ser 370 to Ala. Note that the weak cross-reactivity of the antibodies is due to the similarity in the sequence surrounding Ser329 and Ser370 in CRTC3.
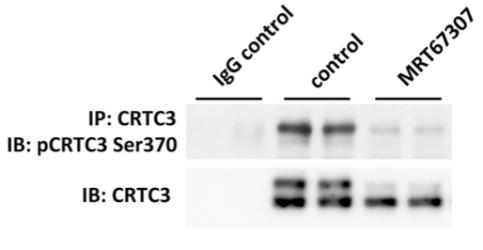
RAW264.7 cells were treated with vehicle control or with different inhibitors of the salt- inducible kinases for 1 hour. Endogenous CRTC3 was immunoprecipitated from 1.5 mg of cell extract protein with 10 ug anti-CRTC3 (S277D, bleed 2). Proteins were separated by SDS-Page and immunoblotted with anti-CRTC3 phospho Ser 370 (S253D) at 125 ng/ml in the presence of 1.25 ug/ml of the non-phospho peptide. Blots were stripped and re-probed using anti-CRTC3 (Abcam cat. Ab91654). The antibody against CRTC3 phospho Ser 370 detects the endogenous CRTC3 after immunoprecipitation from cells that were treated with vehicle control. Phosphorylation is blocked by inhibitors against the salt-inducible kinases such as MRT67307.
PDF Datasheet
CRTC36.5 MB
Sheep No
S253D
Unit Source
Aliquot
0.1mg
Price
£150.00


