Full Name
DAPP1 phospho Tyr 139
Long Name Textual
Dual Adaptor of Phosphotyrosine and 2-Phosphoinositides
Immuno Sequence
PRKVEEPSIY*ESVRVH [residues 130 - 145 of human]
Mono/Poly clonal
Polyclonal
Gel Image
![A solution (9μl) was prepared containing purified recombinant GST-hDAPP1 or GST-hDAPP1 [Y139F] (1μg) and 5mM manganese chloride, 5mM magnesium acetate, 199uM [γ-32P] ATP, 50mM Tris-HCl (pH 7.5) and 0.1% (v/v) β-mercaptoethanol. To this solution was added 1μl of either partially purified Src (1 unit) or Lyn (1 unit) or Sf9 insect lysate expressing Lck (0.2μg) or control Sf9 cell lysate (0.2μg). One unit of enzyme activity was defined by the supplier as that which transfers 1.31 pmoles of phosphate per min using 150μM Src Kinase substrate peptide. The reaction mixtures were then incubated for 30 min at 30ºC in a water bath. The reaction was stopped by the addition of SDS sample buffer to a final concentration of 0.2% (w/v) SDS. The samples were boiled for 5mins and then either all of the sample (A) or 20% of the sample (B) was subjected to SDS-PAGE (7.5% gel). DAPP1](/sites/default/files/styles/gel_image/public/gel_images/DAPP1_Tyr139_S161A_IB_1.jpg?itok=vKFUQREN)
A solution (9μl) was prepared containing purified recombinant GST-hDAPP1 or GST-hDAPP1 [Y139F] (1μg) and 5mM manganese chloride, 5mM magnesium acetate, 199uM [γ-32P] ATP, 50mM Tris-HCl (pH 7.5) and 0.1% (v/v) β-mercaptoethanol. To this solution was added 1μl of either partially purified Src (1 unit) or Lyn (1 unit) or Sf9 insect lysate expressing Lck (0.2μg) or control Sf9 cell lysate (0.2μg). One unit of enzyme activity was defined by the supplier as that which transfers 1.31 pmoles of phosphate per min using 150μM Src Kinase substrate peptide. The reaction mixtures were then incubated for 30 min at 30ºC in a water bath. The reaction was stopped by the addition of SDS sample buffer to a final concentration of 0.2% (w/v) SDS. The samples were boiled for 5mins and then either all of the sample (A) or 20% of the sample (B) was subjected to SDS-PAGE (7.5% gel).
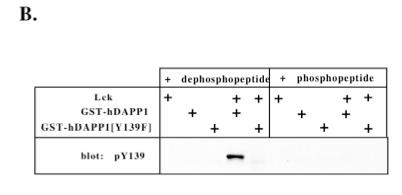
(A) or 20% of the sample (B) was subjected to SDS-PAGE (7.5% gel). (A) The gel was stained with Coomassie Blue and autoradiographed to detect phosphorylated DAPP1. (B) Phosphorylated GST-hDAPP1 was detected by immunoblotting with the affinity purified anti-pY139 antibody (100μg/ml) in the presence of 10μg/ml phosphopeptide immunogen or the equivalent dephosphopeptide.
Sheep No
S161A
Unit Source
Aliquot
0.1mg
Price
£150.00


