Full Name
ATF2 phospho Ser 90
Long Name Textual
Activating Transcription Factor
Immuno Sequence
FNELAS*PFENE [residues 85 - 95 of human]
Mono/Poly clonal
Polyclonal
Gel Image
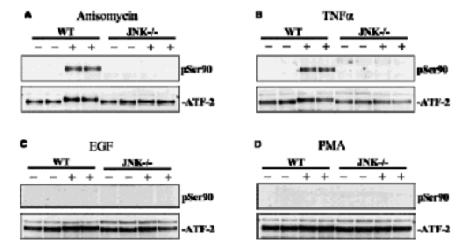
Embryonic fibroblast cells from wildtype (WT) and JNK-/- mice were stimulated for (A) 30 mins with anisomycin (10 μg/ml), (B) for 15 min with TNFalpha (10 ng/ml), (C) for 15 min with EGF (100 ng/ml) and (D) for 30 mins with PMA (100 ng/ml). The cells were lysed and ATF2 was immunoprecipitated from the lysates using 2 μg anti-ATF2 (S512A) per 5 mg of extract. The immunoprecipitate was denatured in SDS, subjected to SDS/PAGE and transferred to nitrocellulose membranes. The membranes were immunoblotted with an antibody that recognises ATF2 phosphorylated at Ser 90 and with an antibody that recognises the phosphorylated and unphosphorylated forms of ATF2 equally well (anti-ATF2 S512A). Binding of the primary antibody was detected using rabbit peroxidase conjugated anti- sheep IgG antibody (1 in 10, 000 dilution, Pierce) followed by enhanced chemiluminescence (ECL, Amersham).
Sheep No
S587A
Unit Source
Aliquot
0.1mg
Price
£150.00


